Decrease in entropy2/15/2024 When the volume decreases, particles come closer and are less random. When the volume increases, particles move far from each other and are more random. Volume: Since pressure and volume are inversely proportional to each other at a given temperature, the entropy relationship also holds inversely.So, entropy increases with a decrease in pressure and decreases with an increase in pressure. In contrast, with the decrease in pressure, particles tend to move away from each other and are less ordered or more random. Pressure: With the increase in pressure, particles come closer to each other, are arranged in a regular pattern, and the system is more ordered.As temperature decreases, particles have less energy, and therefore, move with less speed and are more ordered. As temperature increases, particles gain more energy and move with great speed and the randomness increases. Temperature: With the increase in temperature, entropy increases, and with a decrease in temperature, entropy decreases.So, entropy decreases with an increase in attractive forces. This orderliness decreases from solids to liquids to gases. Particles are placed in a fixed position and are more ordered. Solids have fewer spaces between the particles, so the attractive forces are high. Attractive forces: The attractive intermolecular forces also affect entropy.Thus, solids have the least entropy, liquids have moderate entropy, and gases have the maximum entropy. They need a sealed container to store them. Gases have no fixed shape or fixed volume, and their particles are far from each other. The arrangement of particles is a little less ordered in liquids and it takes the shape of the container it is stored in.  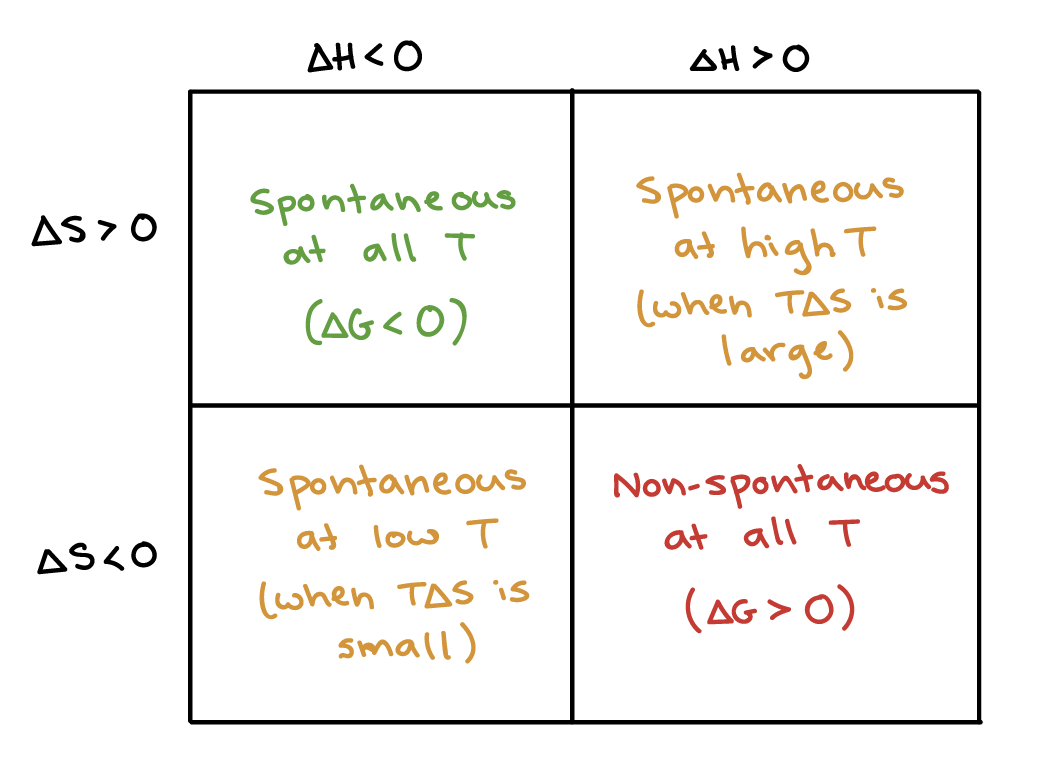
Liquids have no fixed shape but have a fixed volume. Solids have a proper arrangement of particles and have fixed shape and volume, and do not need a container to store them. Characteristics of solids, liquids, and gases are different. Physical state: There are mainly three states of matter – solids, liquids, and gases.Entropy can be variable (high, low or zero), based on the conditions or the thermodynamic system like temperature, pressure, volume etc. Entropy can vary with various factors like physical state, attractive forces, temperature, pressure, volume, molar mass, number of moles. On the other hand, if the system is less ordered, the prediction of the particle behaviour is difficult, and hence the entropy is high. If the system is more ordered, which means that the particles are arranged properly, predicting the movement or behaviour of particles is a little easy. Entropy mainly aims at predicting the direction of the chemical reaction. Sfinal – S initial = ∆S ( ∆ indicates the finite change in the property) What is Entropy?Įntropy is the calculation of the randomness or the disorder present in the molecules or particles of the thermodynamic system. The symbol used for Entropy is S, and the change in Entropy is given as 
So, to find the entropy of a system, only the difference in entropy between the initial and final states can be calculated. 
This is because work can be done only by the ordered motion of particles whereas entropy is the measure of disordered motion of particles. “S” of a system is the measure of thermal energy of the system per unit temperature which is not used (unavailable) to do work. Though they are related, they are different terms. Often the term entropy is confused with energy. Entropy is the measure of the randomness of the particles in a system. A state function is a physical quantity that can be measured by knowing the initial and final values of the system, irrespective of the path followed by it to reach the final state. It is a state function similar to Internal energy or Free energy. The entropy of a system cannot be measured directly but we can find the difference in entropy of the initial and final states of a system.Įntropy is an extensive property that depends on the mass or quantity of matter present in a system. A thermodynamic system is a part of the universe under observation, and the rest of the universe is the surroundings. Entropy is a thermodynamic property that explains the state of a system.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
